Drug delivery
Home > Research > 연구분야 > Drug delivery
1. Stimuli-responsive polymeric nanocarriers (micelles, nanogels) for controlled drug release
Ø Near-infrared light-responsive, diselenide containing
core-cross-linked micelles prepared by the Diels–Alder click reaction for
photocontrollable drug release application
Near-infrared (NIR)
light possesses great advantages for light-responsive controlled drug release,
such as deep tissue penetration and a less detrimental effect on healthy
tissues. In this paper, we report a facile and efficient preparation
of a NIR-triggered micelle system for a drug vehicle. The system consists of
NIR sensitive indocyanine green (ICG) and anticancer drug, doxorubicin (DOX)
which are simultaneously encapsulated in core-cross-linked (CCL) micelles
formed by the Diels–Alder
click reaction between poly (ethylene oxide)-b-poly(furfuryl methacrylate)
(PEO-b-PFMA) and a novel diselenide-containing bismaleimide cross-linker. Under
NIR (808 nm) irradiation, ICG generates reactive oxygen species which can
cleave diselenide bonds in the core of CCL micelles. This response to NIR
irradiation allows the rapid release of DOX from the de-cross-linked micelles,
leading to significantly enhanced
apoptosis in HepG2 cells as evidenced by a cytotoxicity test and confocal laser
scanningicroscopy.

Scheme 1. Preparation of PEO-b-PFMA based CCL micelles and drug release under NIR exposure.
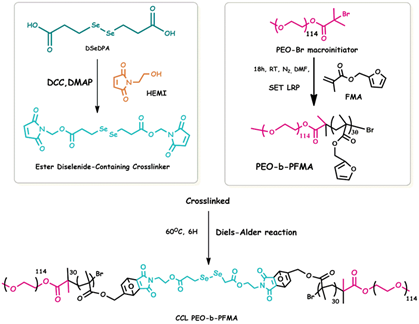
Scheme 2. Synthesis of the ester diselenide cross-linker, PEO-b-PFMA and
CCL micelles
Ref.: Polymer Chemistry 2018, 9, 4813–4823
Ø One-pot
syntheses of dual-responsive core cross-linked polymeric micelles and
covalently entrapped drug by click chemistry
A drug delivery system for
prednisolone 21-acetate (PA) based on dual-responsive core cross-linked (CCL)
micelles was prepared efficiently by alkyne-azide click chemistry. The CCL
micelles of the PEO-b-PGMA-N3 and covalently entrapped PA were prepared
simultaneously using the alkyne-functionalized hydrazone-containing PA and
disulfide-containing crosslinking agent in the presence of CuSO4 5H2O and
sodium ascorbate. The CCL micelles with improved PA loading efficiency (83%)
compared to physically loaded micellar system illustrated the structural
stability of the micelles under physiological condition, while de-cross-linking
through the cleavage of disulfide groups took place rapidly in dithiothreitol
reduction circumstance. In addition, the pH-sensitive hydrolysis of the hydrazone
groups in PA derivative in the micellar core presented a burst release of the
drug at pH 5 and 37 oC in marked contrast to little release at pH
7.4.

Refs: European
Polymer Journal 68
(2016) 264 -273 / European Polymer Journal 68 (2015) 267–277 /
eXPRESS Polymer Letters Vol.11, No.10 (2017) 832–845
Ø Redox-Responsive
Core Cross-Linked Micelles of Poly(Ethylene Oxide)-b-Poly(Furfuryl
Methacrylate) by Diels-Alder Reaction for Doxorubicin Release
Redox-responsive core cross-linked (CCL)
micelles of poly(ethylene oxide)-b-poly(furfuryl methacrylate) (PEO-b-PFMA)
block copolymers were prepared by the Diels-Alder click-type reaction. MTT assays demonstrated
that the block copolymer did not have any practically cytotoxicity against the
normal HEK293 cell line while DOX-loaded CCL micelles exhibited a high
antitumor activity towards HepG2 cells

Refs : JOURNAL OF POLYMER SCIENCE, PART A: POLYMER CHEMISTRY 2016, 54, 3741–3750 / eXPRESS Polymer Letters Vol.12, No.8 (2018) 688–698
2.
Synthesis of biodegradable
hydrogels for injectable implants
Ø
Near-Infrared Light-Responsive Hydrogels for on
Demand Drug Release
Our research group developed a hydrogel platform, which
has an ability to be triggered by near infrared (NIR) light. The presence of
inverse electron demand Diels-Alder (IEDDA) groups in the hydrogels leads a
fast gelation time in a mild condition without any catalyst. By employing
sensitive cross-linkers having Se-Se bonds, we demonstrate a gel-sol transition
of the hydrogels under NIR irradiation. With the ratio of precursors, we could
manipulate the degradation of the hydrogels. The in vitro degradation
and drug release studies reveal that NIR light triggers the de-cross-linking of
hydrogels and release of loaded drugs.

Scheme 1. Schematic illustration of NIR-responsive hydrogels.
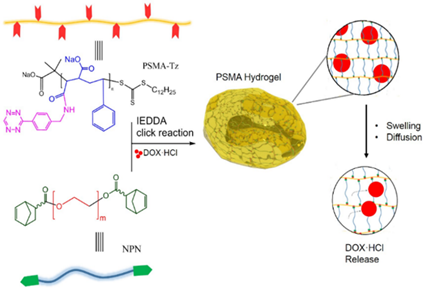
Ø
Click-cross-linked
Injectable Hydrogels Based on Poly(styrene-alt-maleic anhydride) for Controlled
Doxorubicin Release
We synthesize a novel injectable hydrogel based on
poly(styrene-alt-maleic anhy-dride) (PSMA) via inverse electron demand
Diels-Alder click chemistry. The hydrogel is formed rapidly (within several
minutes) by the click-cross-linking reaction of PSMA-3-(p-benzylamino)-1.2.4.5-tetrazine
and poly(ethylene glycol)-5-norbornene-2-carboxylic acid. The precursors
exhibit practically no depressing effect on the normal HEK293 cell growth while
the DOX•HCl-loaded hydrogel presents significant anticancer activity towards
HeLa cells, which makes this biocompatible hydrogel potential for controlled
anticancer drug release.